Popular Articles
- Earliest molecular events of vision revealed
- Dynamics and Kinetics in Structural Biology
- Structural biology is solved -- now what?
- XFEL Pulses Demonstrate How Plants Perceive Light
- BioXFEL Postdoctoral Fellowship Award
Archived Articles
- Details
- Thursday, 27 July 2017

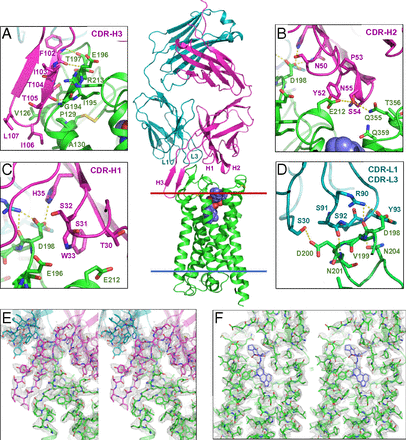
Monoclonal antibodies provide an attractive alternative to small-molecule therapies for a wide range of diseases. Given the importance of G protein-coupled receptors (GPCRs) as pharmaceutical targets, there has been an immense interest in developing therapeutic monoclonal antibodies that act on GPCRs.
Here we present the 3.0-Å resolution structure of a complex between the human 5-hydroxytryptamine 2B (5-HT2B) receptor and an antibody Fab fragment bound to the extracellular side of the receptor, determined by serial femtosecond crystallography with an X-ray free-electron laser. The antibody binds to a 3D epitope of the receptor that includes all three extracellular loops. The 5-HT2B receptor is captured in a well-defined active-like state, most likely stabilized by the crystal lattice. The structure of the complex sheds light on the mechanism of selectivity in extracellular recognition of GPCRs by monoclonal antibodies.





