Popular Articles
- Earliest molecular events of vision revealed
- Dynamics and Kinetics in Structural Biology
- Structural biology is solved -- now what?
- XFEL Pulses Demonstrate How Plants Perceive Light
- BioXFEL Postdoctoral Fellowship Award
Archived Articles
- Details
- Thursday, 10 August 2017

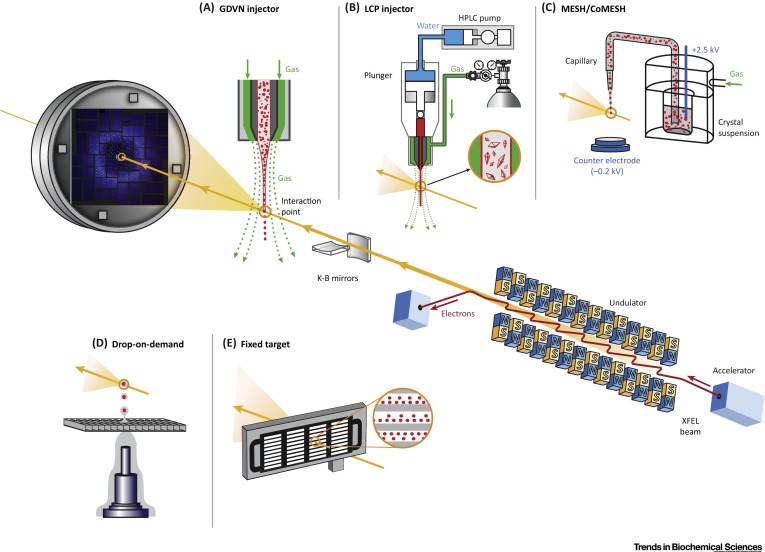
BioXFEL scientist Vadim Cherezov and his colleagues released a new publication to CellPress:
X-ray free electron lasers (XFELs) have the potential to revolutionize macromolecular structural biology due to the unique combination of spatial coherence, extreme peak brilliance, and short duration of X-ray pulses. A recently emerged serial femtosecond (fs) crystallography (SFX) approach using XFEL radiation overcomes some of the biggest hurdles of traditional crystallography related to radiation damage through the diffraction-before-destruction principle.
Intense fs XFEL pulses enable high-resolution room-temperature structure determination of difficult-to-crystallize biological macromolecules, while simultaneously opening a new era of time-resolved structural studies. Here, we review the latest developments in instrumentation, sample delivery, data analysis, crystallization methods, and applications of SFX to important biological questions, and conclude with brief insights into the bright future of structural biology using XFELs.





